BOC Protection and Coupling of L-Phenylalanine for Antimicrobial Testing
Location
CoLab, COM 262
Start Date
30-4-2026 12:00 PM
Document Type
Poster
Description
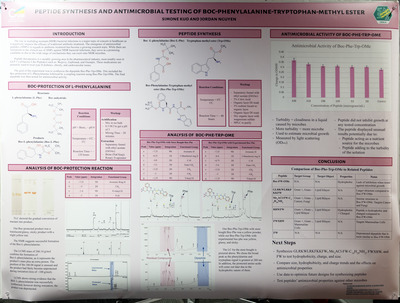
In this experiment, a dipeptide was synthesized using L-phenylalanine as the starting amino acid. The amino group of L-phenylalanine was first protected with a tert-butoxycarbonyl (Boc) group, which leaves the carboxylic acid free to participate in a peptide coupling reaction. After formation of the peptide bond through the coupling reaction, the Boc group was removed to regenerate the free amine on the dipeptide. Reaction progress was monitored using thin-layer chromatography (TLC), and the final product was purified to remove any remaining reagents and by-products. The structure and purity of the synthesized dipeptide were analyzed using NMR spectroscopy, mass spectrometry, and LC-MS. Finally, the purified dipeptide was tested for antimicrobial activity to evaluate its potential biological properties.
BOC Protection and Coupling of L-Phenylalanine for Antimicrobial Testing
CoLab, COM 262
In this experiment, a dipeptide was synthesized using L-phenylalanine as the starting amino acid. The amino group of L-phenylalanine was first protected with a tert-butoxycarbonyl (Boc) group, which leaves the carboxylic acid free to participate in a peptide coupling reaction. After formation of the peptide bond through the coupling reaction, the Boc group was removed to regenerate the free amine on the dipeptide. Reaction progress was monitored using thin-layer chromatography (TLC), and the final product was purified to remove any remaining reagents and by-products. The structure and purity of the synthesized dipeptide were analyzed using NMR spectroscopy, mass spectrometry, and LC-MS. Finally, the purified dipeptide was tested for antimicrobial activity to evaluate its potential biological properties.

Comments
The faculty mentor for this project was Meagan Weldele.