Peptide Synthesis
Location
CoLab, COM 217
Start Date
30-4-2026 10:45 AM
Document Type
Poster
Description
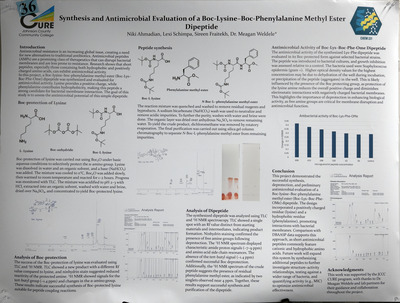
Antimicrobial resistance remains a critical global health challenge, with millions of infections occurring annually in the United States alone. Peptide-based therapeutics represent a promising alternative to traditional antibiotics. In this project, we aimed to synthesize a dipeptide in the liquid phase using a coupling reagent to join amino acids together with the use of protecting groups. These protecting groups are necessary to ensure selective formation of the desired peptide bond. The synthesized peptide will be tested for antimicrobial activity and compared to other active peptides. Peptide bond formation involved the addition and subsequent removal of protecting groups, and the products were analyzed for structure and purity. The dipeptide was purified and tested for antimicrobial activity.
Peptide Synthesis
CoLab, COM 217
Antimicrobial resistance remains a critical global health challenge, with millions of infections occurring annually in the United States alone. Peptide-based therapeutics represent a promising alternative to traditional antibiotics. In this project, we aimed to synthesize a dipeptide in the liquid phase using a coupling reagent to join amino acids together with the use of protecting groups. These protecting groups are necessary to ensure selective formation of the desired peptide bond. The synthesized peptide will be tested for antimicrobial activity and compared to other active peptides. Peptide bond formation involved the addition and subsequent removal of protecting groups, and the products were analyzed for structure and purity. The dipeptide was purified and tested for antimicrobial activity.

Comments
The faculty mentor for this project was Meagan Weldele.